(CMR) Mead Johnson Nutrition (MJN) has recalled select batches of Nutramigen Powder due to a possibility of contamination with Cronobacter sakazakii bacteria in products sampled outside the United States, the Food and Drug Administration said.
According to Reuters, the FDA said Nutramigen Powder, a specialty infant formula for the dietary management of Cows Milk Allergy (CMA) in 12.6- and 19.8-ounce cans, went through extensive testing by MJN and tested negative for the bacteria.
The Nutramigen containers were manufactured in June 2023 and distributed primarily in June, July, and August.
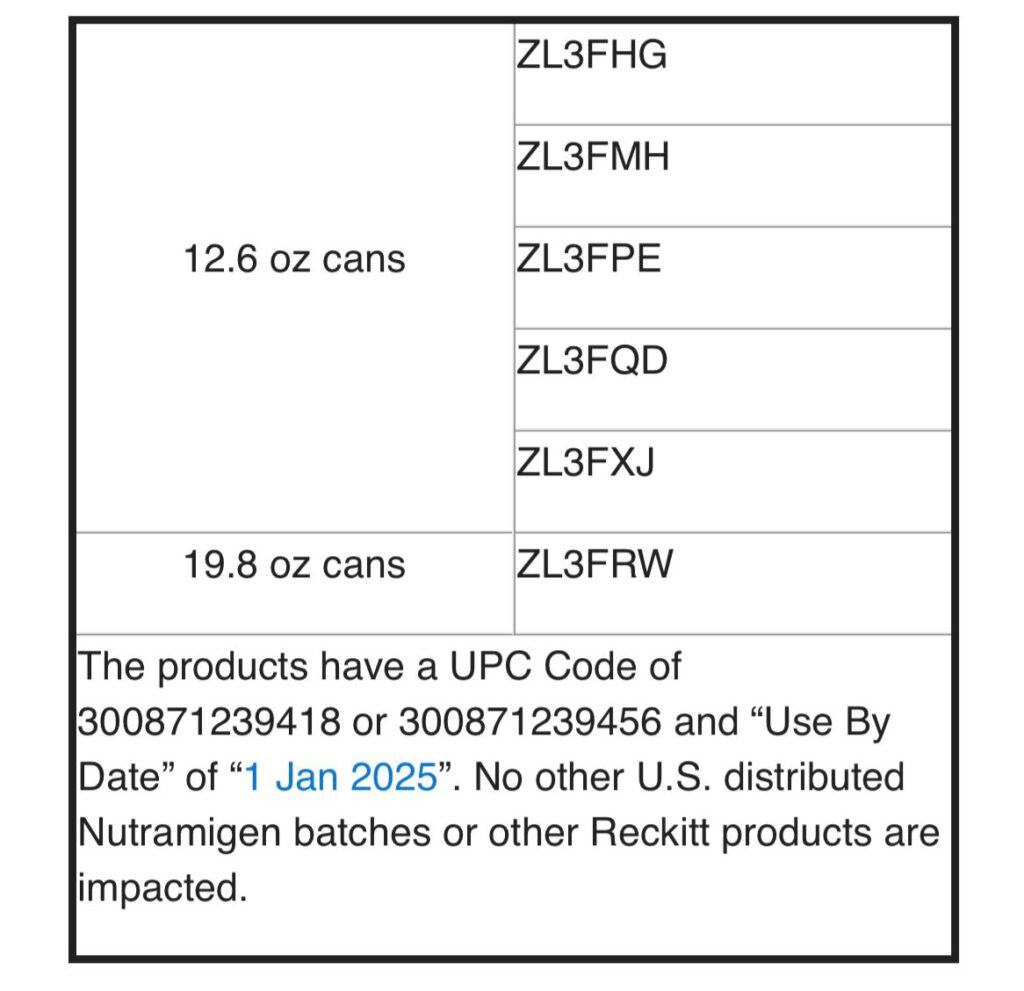
“Based on the limited availability of the remaining stock of this special infant formula, it is believed that much, if not all, of the products recalled in the United States, have been consumed,” the FDA said, adding there are no reports of illnesses or adverse events to date.
Reckitt Benckiser Group, under which MJN falls, said all the recalled products have the “Use By Date” of “1 Jan 2025,”and customers are to dispose of the products if they belong to the recalled batches.
“No other U.S.-distributed Nutramigen batches or other Reckitt products are impacted,” Reckitt said.
According to the CDC, cronobacter sakazakii infection is rare but can be especially dangerous for babies under 2 months, premature babies and babies with weakened immune systems, as well as older adults over the age of 65. Cronobacter bacteria can cause severe, life-threatening infections or meningitis. The infection may also cause bowel damage and may spread through the blood to other parts of the body.
- Fascinated
- Happy
- Sad
- Angry
- Bored
- Afraid